 So I saw electronics is, um, ions or elements or are Adams? Could be. So really, you can see me on with 10 electrons, really matches the other ions here. And that would be our 10 electrons, which would give us a minus three to confirm. So let's seven protons here, okay? And if you had this minus three now you can see from the oxygen example that would be tomb or electrons on again. Maybe the nitride I on with a minus three year again, Nitrogen is gonna have atomic number of seven. You want to match that charge? You can see how these ions with the charges. Right? So that you end up with a plus two, which would match again. 
So how many electrons would you need? Here and again? That would be 10. If you have 12 protons, the most common ay on off magnesium is typically plus two. We could say, OK, that would be 12 protons. How about magnesium? So it's magnesium atomic numbers. We could move around me on the periodic table and find some other elements that will be close by. You could see that you have 11 protons for a positive 11 and the same matching 10 electrons. What, without charge B, in this case, well, we'll be looking at really quickly. And let's see if we can come up with our our charge. You'd have 11 proton in this case, right? So if you looked at the same number of electrons me on, you would have 10 electrons again. If you want a sodium, we'll see the atomic number of sodium is gonna be 11. If we go over to sodium, let's try that one. 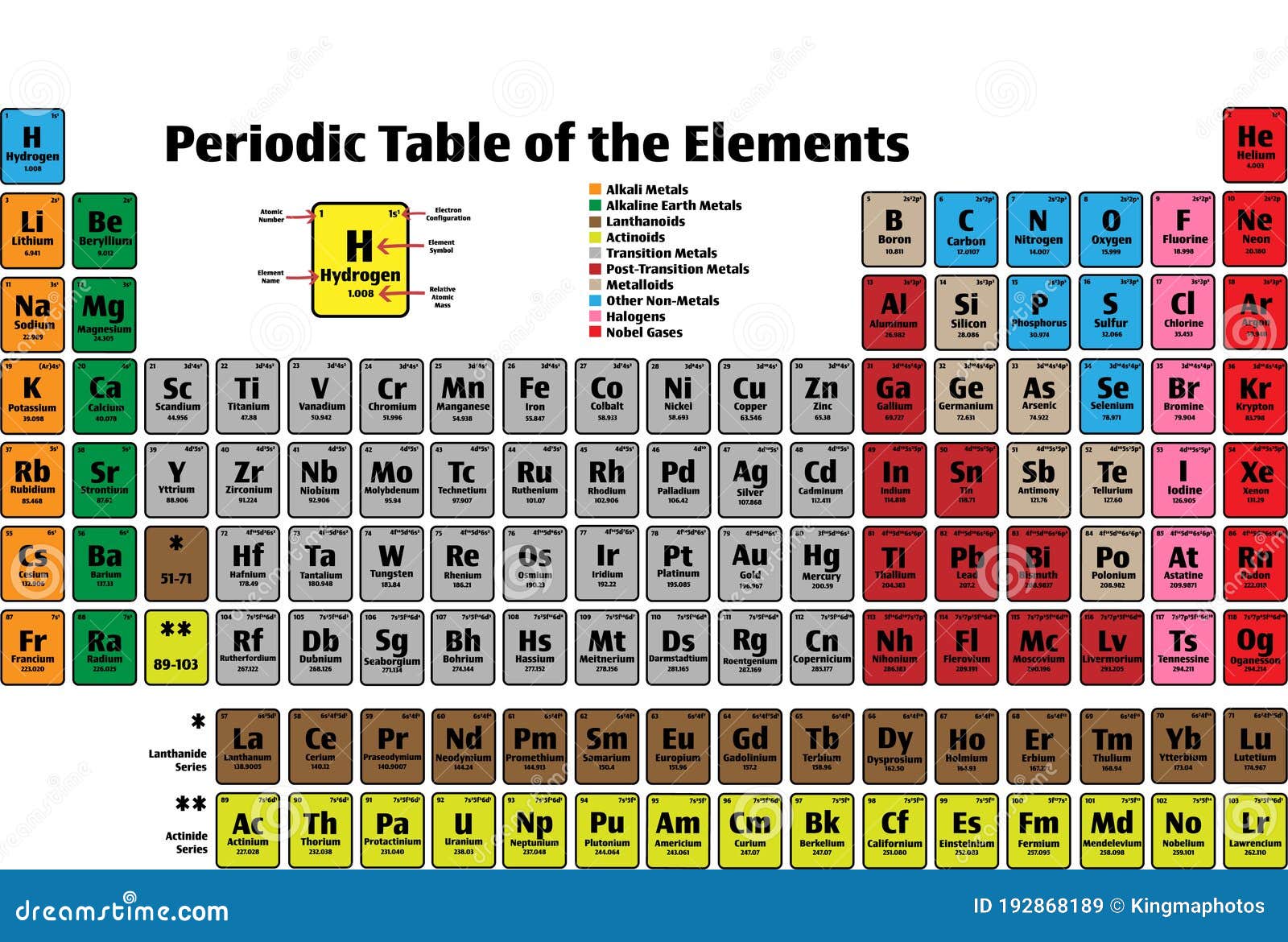
And you'll see that matches the charge on the ion of oxygen to the oxide island. Okay, so what we're gonna do is that we add two electrons that becomes 10 electrons that would now have a minus two charge. So I have eight protons for a positive eight and eight electrons. Well, oxygen is gonna have an atomic number of eat, so that would be eight protons. So if we choose something like the oxide, I would say, Oh, with a to minus, we'll see how we develop this. That's gonna mean that you have a charge or miss balance of electrons and protons. Dio is we want to find ions with charges that would have the same number of electrons as neon neon Adam would be which be 10. So together, if you and those together you'll cancel in both down you get a zero charge. We see the neon as atomic number of 10 so it's gonna 10 protons will also have 10 electrons. So when we haven't Adam, that simply means that the protons and electrons gonna be the same. His neon is gonna have a zero charge for an Adam of neon. We'll see that neon we draw it out real quick. So let's just look at me on really quickly. Atomic number is an important concept to learn and is significant in understanding the trends of the periodic table and later in chemical reactions as well.Okay, everyone, today we're gonna take a look at Neon, and we want to find four ions that have the same number of electrons as neon. However, having basic knowledge of the periodic table and the characteristics of elements will help you answer this. Such questions can be tricky and require you to know the definitions of such terms. Therefore, its atomic number is also 26 in the periodic table. The elements in the periodic table have been arranged according to their atomic number.įor example, an atom of the element iron has 26 protons in its nucleus, which implies that it has 26 electrons. The number of electrons in an atom of an element is equal to its atomic number. Therefore, since the number of protons in an element is equal to the number of electrons – option (D) is the correct answer. Option (B) atomic number of a chemical element is defined as the number of protons found in the nucleus of every atom of that element. With this definition, option (D) can also be ruled out. It depends on the atomic size, effective nuclear charge, and reactivity of elements as well. Option (D) electron affinity is defined as the potential energy change or the amount of energy released when an electron is added to a neutral gaseous atom, forming an anion. With this definition, option (C) can also be ruled out. 
This concept has been displaced by the concept of molar mass. Option (C) equivalent weight is defined as the quantity of a substance that exactly reacts with, or is equal to the combining value of, an arbitrary fixed quantity of another substance in a particular reaction. With this definition, we can rule out options (A). Option (A) atomic weight is the total weight of the atom, which includes the weight of the protons and neutrons and the little extra weight of electrons as well. With this information, and definitions for each of the above options, the answer can be found. Hint :The number of electrons in an atom of an element is equal to the number of protons in the atom. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
